Should you’ve grow to be unplugged from the evolving headlines surrounding Elon Musk’s brain-computer interface (BCI) startup, Neuralink, right here’s a recap:
- In January, Neuralink implanted their first participant, Noland Arbaugh, with the N1 implant at Barrow Neurological Institute in Phoenix, AZ. Based on Neuralink’s web site, “The N1 Implant data neural exercise via 1024 electrodes distributed throughout 64 threads. These highly-flexible, ultra-thin threads are key to reduce injury throughout implantation and past.”
- In mid-March, Noland was introduced to the world by way of livestream by Neuralink (on Musk’s platform, X), taking part in video games utilizing his implant.
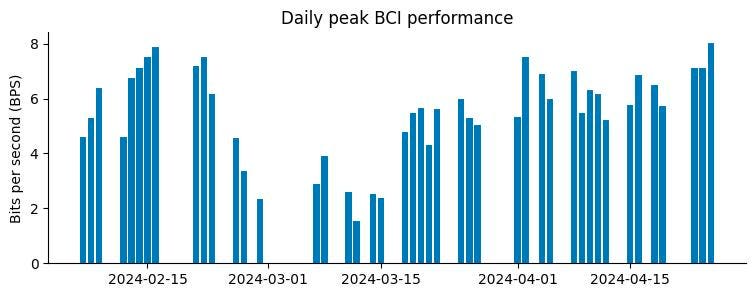
From Neuralink’s Weblog “Every day peak efficiency in bits per second (BPS)”
- On Might 8, an organization weblog submit revealed that within the weeks following the surgical procedure, “a variety of threads retracted from the mind, leading to a web lower within the variety of efficient electrodes.” (see dip within the chart above.) Regardless of this malfunction, the corporate was capable of restore full perform via augmenting software program, with no need further surgical correction of the implant’s wires.
- The subsequent day, The Wall Avenue Journal ran a story, speculating attainable the explanation why the implant might need malfunctioned, and suggesting that it was their preliminary inquiries to Neuralink that led to publication of the weblog submit. A Wired piece the identical day explained numerous mechanical variations in at present’s commercially oriented (however pre-market) BCI units, citing exterior opponents and consultants who pointed to the significance of sturdiness as a key design attribute on this know-how.
- On Might 15, Reuters reported that Neuralink has recognized in regards to the wires’ retraction points for years, going again to their animal testing section, citing 5 nameless sources talking behind confidentiality agreements. Certainly one of these sources mentioned that the FDA’s preliminary rejection of Neuralink’s utility to start human trials included security issues in regards to the implant’s threads.
- On Might 20, one other WSJ article reported that 85% of the threads had retracted, that the subsequent implant shall be implanted 8 millimeters deep (in comparison with 3 to five millimeters in Arbaugh), and that the FDA had greenlit Neuralink’s recruitment for his or her second affected person.
- On Might twenty second, Arbaugh spoke with Wired again to share his perspective. When requested whether or not he’d recognized that wire retraction was attainable, he mentioned, “I didn’t have any information that it was attainable. I don’t assume they noticed it in any of the animal trials. I had heard that it had possibly occurred in one of many monkeys however that it was a lot completely different. It was by no means anticipated that it might occur in me.” He later said, “It looks like the threads have stabilized, and even some that had been pulled out of my mind had discovered their manner again in.”
Since then: Extra interviews, extra media protection, extra hypothesis, extra questions. With Musk there’s at all times extra. However let’s cease the tape right here and mirror.
Within the span of two weeks: One firm weblog submit, 5 nameless sources, 4 nice reporting groups (amongst many others), one sociable participant and one silent federal company have turned this story – a few security trial for a medical system – right into a web-wide drama with extra questions than solutions.
A very powerful element: Arbaugh seems to be okay. However as a substitute of transferring on, the neurotech business would possibly take into account this a teachable second for reflection.
The query isn’t “What went fallacious?” however “What will be realized from this?”
A caveat: The retraction challenge that occurred is predicated on Neuralink’s particular product design, whereby the threads proven under got here free. Different firms’ BCIs use completely different technique of implanting electrodes onto or into the mind.
How can the stewards of BCI’s fast-approaching future use this episode to enhance security, optimize entry and strengthen the general public’s belief on this know-how?
Neuralink’s N1 implant
What Can Neuralink Do Otherwise Transferring Ahead?
No person expects a lot to alter. Musk has a well-documented mistrust and disdain for each media and the federal government, and a penchant for controlling the narrative. However as an organization, Neuralink has proven a glimmer of willingness to respond to media and regulatory criticism on their weblog, albeit not directly, after the actual fact and stopping effectively in need of a mea culpa.
I could also be within the minority right here however I assumed their Might 8 weblog submit was an excellent first response: direct, succinct, sensible. A technical resolution to a technical drawback.
The difficulty is that it didn’t go far sufficient. BCI are usually not nuts and bolts. They’re a human-powered know-how. This can be a human trial. Arbaugh is a human being.
So, past the mechanical resolution of deeper implants, what did they be taught from this incident, their very first involving an precise particular person? What modifications are they exploring to enhance subsequent human trials? How will they evolve to reside as much as their stated tradition, the place “open communication and collaboration underpin our dedication to transparency and championing the perfect concepts, no matter the place they arrive from” ?
As a substitute of hazarding such self-awareness, Neuralink has remained characteristically opaque on the matter at hand to date, abstaining from media feedback and diverting consideration as a substitute in direction of their recruitment of a second US affected person, advances in world trials and ongoing product improvement associated to restoring imaginative and prescient and contact by way of BCI.
The corporate has additionally issued a brand new “technical challenge” whose handy timing will assist shift the narrative. And so it goes.
However it’s not too late. In reality it’s nonetheless fairly early. Let’s keep watch over that blog.
Can We Enhance Knowledgeable Consent for Experimental Trials?
Arbaugh clearly didn’t perceive retraction was a threat. Neuralink clearly did.
The actual particulars of Arbaugh’s pre-trial onboarding will probably stay non-public. However what will be achieved to enhance this sort of info asymmetry for different individuals?
These enrolling in new BCI trials are going the place none (or few) have gone earlier than. Knowledgeable consent requires that contributors are notified of any “fairly foreseeable dangers.”
Far past BCI, knowledgeable consent is an even bigger can of worms than the Dune sequel.
It’s ripe for reform: “the topic of debate for no less than 3 a long time, with a constant consensus all through this time that these paperwork are too tough to learn, too advanced, and too lengthy and, because of this, incessantly fail to facilitate really knowledgeable consent by examine contributors.”
Knowledgeable consent isn’t overseen by the FDA (which seemed to know in regards to the threat of wire retraction in N1 implants,) however by a completely separate workplace inside HHS referred to as the Workplace of Human Analysis Safety. The OHRP chooses when to train regulatory oversight. However STAT’s current crackerjack reporting on an unrelated and ongoing knowledgeable consent debacle at Mt. Sinai, confirmed how this workplace merely ignored the FDA’s e-mail “alarm bells,” regardless of repeated breakdowns in defending sufferers from medically doubtful mind biopsies that one neurosurgeon described as “nearly Geneva Conference stuff.”
Within the years forward, as firms develop and take a look at BCIs, every with completely different system designs and implantation protocols, brain-related research may grow to be a textbook instance of the pressing want to enhance the knowledgeable consent course of on the federal degree to guard individuals’s most inviolable core of autonomy, identification, and consciousness. Incorporating accessible language appropriate for each schooling degree, new formats and tools and higher avenues to ask questions could guarantee extra dangers are understood with out the necessity for an legal professional.
To be clear: This goes far past Neuralink or any BCI firm. Whereas trial contributors aren’t fairly flying blind, enrolling in trials for experimental mind implantation represents a fancy alternative amidst strongly vested pursuits. As Arbaugh put it:
“…I’m a quadriplegic, and all I actually have is my mind. So letting somebody go in there and fiddle, it’s a giant dedication. If one thing goes fallacious, that’s sort of it for me. However I knew I needed to assist out, and I didn’t need to let my fears get in the way in which of that. The second factor that gave me pause was that I didn’t know if I needed to be the primary one to get this in my mind if something would go fallacious with the implant. What if it breaks or stops working and I solely have it for a day, every week? I assumed possibly another person ought to get it first, and I’ll get the higher model of it.”
As per his needs above, how can subsequent sufferers in every scientific trial be higher knowledgeable by the expertise of the primary? The BCI Pioneers Coalition exists to make sure these in Arbaugh’s place don’t need to shoulder the accountability of training new trial contributors about dangers and advantages of an investigational examine. However not all contributors in BCI trials are conscious of this useful resource, or different avenues of assist, earlier than they be a part of a trial.
What Counts as “Promotion” underneath IDE?
As per the BPS graph above: On March 20, when Neuralink launched the world to Arbaugh by way of Musk’s personal social media platform, they’d recognized in regards to the wire challenge for barely two weeks. But, somewhat than pausing, reflecting, monitoring, they started filming.
It’s not extraordinary for the primary affected person in an FDA-regulated security trial to grow to be a PR venture by the trial sponsor. However why doesn’t this habits depend as “promotion” underneath the FDA’s guidelines for Investigational Gadget Exemption (IDE) research?
As per the FDA’s IDE guidelines, “Sec. 812.7 Prohibition of promotion and different practices. A sponsor, investigator, or any particular person performing for or on behalf of a sponsor or investigator shall not: (a) Promote or take a look at market an investigational system, till after FDA has accepted the system for industrial distribution.”
The FDA is working in a pre-social media world, a pre-Elon-Musk-world, and a pre-Elon-Musk-owning-social-media-world.
To be honest, the company isn’t accountable for anticipating these sorts of unprecedented conditions or traditionally highly effective actors. However now that it has occurred – continues to be occurring – it’s now not unprecedented. Is that this a regulation, or a suggestion?
Company management is legally required to “decline to remark” on such issues, besides: How, when, or will they make clear, adapt or implement their insurance policies?
Is Selling BCI Actually a Affected person’s Job?
Arbaugh has been admirably vocal about his want to contribute to advancing BCI. By all indications, Neuralink has been there for him and his household in main methods, earlier than, throughout, and after implantation. However absolutely the mixed function of extremely seen spokesman and trial analysis topic is loads of strain for one particular person.
The affected person advocates I’ve spoken to are in alignment that consciousness and schooling – significantly in partaking most people – are a key precedence for BCI. Dystopian ‘thoughts management’ narratives don’t mirror actuality; the story wants correcting.
However first comes security, then comes advocacy.
A sensible query for the years forward: Is there a distinction between selling or take a look at advertising an investigational system, and recruiting for scientific trials and affected person registries, vis-a-vis devoted web sites, social media, and past?
Neuralink, whose registry has reportedly recruited greater than 1,000 sufferers, is now increasing their trials into Canada and Britain. Arbaugh’s phrases and likeness blanket the web site. He appears glad to function unpaid ambassador, regardless of the centripetal forces of a media carousel that advantages the corporate and their ongoing trial way over him.
This gray space isn’t fully distinctive to Neuralink. BCI exists within the public creativeness already. Startups are wading into this as they commercialize.
Rival Synchron, who launched their registry this 12 months in preparation for a larger scale clinical trial, has additionally shared their contributors’ tales on nationwide tv and social media, albeit with out fixing a everlasting highlight onto them. Each firm taking BCI via the FDA might want to recruit sufferers and, to an extent, construct belief from the general public.
As neurotechnology climbs the hype cycle, one can neglect that “BCI” are usually not only a trending subject, however medical units designed to stay inside individuals’s our bodies for years. The “moral entanglements” between analysis groups and trial contributors don’t cease on the finish of those multi-year trials. Concerns for the long tail that follows – ongoing scientific care tips, public-private responsibilities to sufferers and the financial implications for protection and reimbursement – are simply rising.
Because the FDA’s new implantable BCI collaborative community convenes this 12 months, affected person advocacy teams will thankfully be on the desk to information the sector. Past public schooling and messaging, their management shall be essential to stability company pursuits in model constructing, scientific pursuits in trial recruitment, and regulatory pursuits in advancing revolutionary therapies safely.
Good Information for Individuals Who Love Dangerous Information
When essentially the most highly effective non-public and public stakeholders concerned in a narrative don’t remark, some individuals begin talking off the report, others speculate, and a few like Arbaugh choose up the slack and take it on themselves.
Leaders in my neurotech neighborhood are in energetic dialogue about how finest to speak about such extremely technical, highly-regulated, still-developing information with media and most people.
Regardless of the musk of intrigue and drama, I imagine this story is sweet information. Right here’s why.
- Noland Arbaugh is okay. Regardless of the wire retraction, his BCI works nice, his sponsor has confirmed adaptive and inventive, and he’s nonetheless capable of do what he enjoys. Together with his optimism and beauty underneath strain he has dealt with this example admirably. I hope he doesn’t burn himself out with media. I’d gently recommend that discovering assist outside of Neuralink is sweet thought.
- If this a lot essential dialogue about BCI can emerge from a single story, due to a mix of sharp reporting, prepared confidants, and sure, a polarizing superstar billionaire founder, that may be a nice end result. Hopefully all stakeholders are capable of take up some classes and implement some modifications – even when they don’t need to (or can’t legally) speak about it.
- This bodes effectively for different BCI firms, current and future, as a reminder to prioritize methodical and prudent compliance with security research over pace to market and public optics. In the long run, this underscores that sufferers will want a number of choices in order that they aren’t pressured to decide on one product (or registry.) Buyers ought to really feel assured on this stage of BCI’s regulatory journey, even when income stays years away.
- Neuralink has seemingly realized essential product optimization and made changes on the fly. That is what profitable startups do. Hopefully, there are not any further malfunctions involving human topics. Let’s want them luck, whereas holding them to the next customary of excellence in scientific trials as a extremely seen chief on this nascent subject.